SoapySouter
Member
- Joined
- Jan 29, 2014
- Messages
- 14
- Reaction score
- 18
Hi, looking for some help. I made liquid soap for the second time yesterday. Last time it went cloudy and runny when I added the fragrance, but was acceptable for home use.
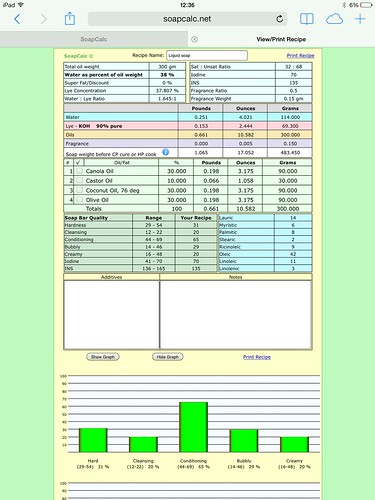
This time, I went for 5% excess KOH (with citric acid as back up) and got a lovely thick, clear soap. I then added the fragrance to the thickly diluted soap, after heating the soap back up and the whole thing turned back into soapy mashed POTATOES!
I'm letting it cools and it's separating...
Can anyone advise- can this soap be saved?
Is it because it's lye heavy and it's saponifying the fragrance oil perhaps
Any help would be greatly appreciated...
(I used 50/50 water/glycerine method)
This time, I went for 5% excess KOH (with citric acid as back up) and got a lovely thick, clear soap. I then added the fragrance to the thickly diluted soap, after heating the soap back up and the whole thing turned back into soapy mashed POTATOES!
I'm letting it cools and it's separating...
Can anyone advise- can this soap be saved?
Is it because it's lye heavy and it's saponifying the fragrance oil perhaps
Any help would be greatly appreciated...
(I used 50/50 water/glycerine method)